Downloads
Download


This work is licensed under a Creative Commons Attribution 4.0 International License.
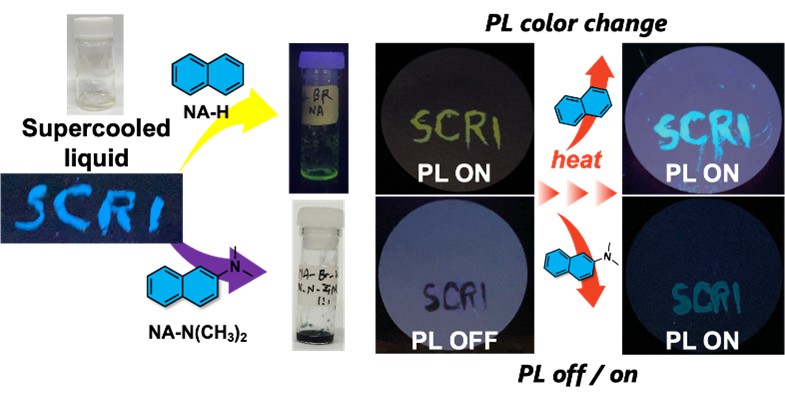
Stimuli-responsive luminescent liquid materials have recently attracted considerable attention due to their potential to address the limitations of solid-state materials, such as the necessity of organic solvents and the difficulty in fabricating composite systems. Liquid-state materials offer superior processability and enable facile modulation of photophysical properties by simply selecting appropriate solutes. In particular, molecular designs incorporating electron-donating or electron-accepting properties into liquid materials allow to form charge-transfer (CT) complexes upon dissolving solutes with their opposite electronic properties, altering both solution color and photoluminescence (PL) behavior. In this study, we developed a room-temperature supercooled liquid material based on an electron-accepting naphthalene diimide (NADI) derivative, BR-Val-NADI. Upon dissolving electron-rich naphthalene-based derivatives (NA-##s) into BR-Val-NADI, NA-##/BR-Val-NADI with CT character were readily obtained as solutions, exhibiting various colors and PL properties. NA-##/BR-Val-NADI also functioned as printable PL inks that could be applied onto various substrates such as glass and paper. Notably, the PL properties of NA-##/BR-Val-NADI were responsive to thermal stimuli, with temperature-induced changes in PL color and PL off/on switching. These results highlight the potential of NA-##/BR-Val-NADI as a new class of stimuli-responsive soft materials for applications in printable photonic devices and smart sensing platforms.
Keywords:
naphthalene diimide stimuli response liquid material stimuli-responsive liquid photoluminescenceReferences
- Yerushalmi, R.; Scherz, A.; van der Boom, M.E.; Kraatz, H.-B. Stimuli responsive materials: New avenues toward smart organic devices. J. Mater. Chem. 2005, 15, 4480–4487. https://doi.org/10.1039/B505212B.
- Huang, Y.; Ning, L.; Zhang, X.; Zhou, Q.; Gong, Q.; Zhang, Q. Stimuli-fluorochromic smart organic materials. Chem. Soc. Rev. 2024, 53, 1090–1166. https://doi.org/10.1039/D2CS00976E.
- Yan, D.; Wang, Z.; Zhang, Z. Stimuli-Responsive Crystalline Smart Materials: From Rational Design and Fabrication to Applications. Acc. Chem. Res. 2022, 55, 1047–1058. https://doi.org/10.1021/acs.accounts.2c00027.
- Sun, H.; Shen, S.; Li, C.; Yu, W.; Xie, Q.; Wu, D.; Zhu, L. Stimuli-Responsive Dual-Emission Property of Single-Luminophore-Based Materials. Adv. Funct. Mater. 2025, 35, 2415400. https://doi.org/10.1002/adfm.202415400.
- Wu, W.; Chen, K.; Wang, T.; Wang, N.; Huang, X.; Zhou, L.; Wang, Z.; Hao, H. Stimuli-responsive flexible organic crystals. J. Mater. Chem. C 2023, 11, 2026–2052. https://doi.org/10.1039/D2TC04642C.
- Huang, L.; Qian, C.; Ma, Z. Stimuli-Responsive Purely Organic Room-Temperature Phosphorescence Materials. Chem.-Eur. J. 2020, 26, 11914–11930. https://doi.org/10.1002/chem.202000526.
- Karunakar, K.K.; Cheriyan, B.V.; Anandakumar, R.; Murugathirumal, A.; Kataria, K.; Yabase, L. Stimuli-Responsive Smart Materials: Bridging the Gap Between Biotechnology and Regenerative Medicine. Bioprinting 2025, 48, e00415. https://doi.org/10.1016/j.bprint.2025.e00415.
- Zhao, J.; Du, J.; Qin, T.; Zhang, S.X.-A.; Sheng, L. “Confined Eutectic” Strategy for Visual Refrigeration Responsive Fluorescent Materials with Easy Preparation and Multi-Color Tunability. Adv. Sci. 2025, 2503779. https://doi.org/10.1002/advs.202503779.
- Du, J.; Sheng, L.; Xu, Y.; Chen, Q.; Gu, C.; Li, M.; Zhang, S.X.-A. Printable Off-On Thermoswitchable Fluorescent Materials for Programmable Thermally Controlled Full-Color Displays and Multiple Encryption. Adv. Mater. 2021, 33, 2008055. https://doi.org/10.1002/adma.202008055.
- Du, J.; Sheng, L.; Chen, Q.; Xu, Y.; Li, W.; Wang, X.; Li, M.; Zhang, S.X.-A. Simple and general platform for highly adjustable thermochromic fluorescent materials and multi-feasible applications. Mater. Horiz. 2019, 6, 1654–1662. https://doi.org/10.1039/C9MH00253G.
- Davey, R.J.; Schroeder, S.L.M.; ter Horst, J.H. Nucleation of Organic Crystals—A Molecular Perspective. Angew. Chem. Int. Ed. 2013, 52, 2166–2179. https://doi.org/10.1002/anie.201204824.
- Liang, C. Organic Polymorphs Based on AIE-Active Molecules: Preparation, Characterization, and Application. Cryst. Growth Des. 2024, 24, 7322–7341. https://doi.org/10.1021/acs.cgd.4c00499.
- Chung, H.; Diao, Y. Polymorphism as an emerging design strategy for high performance organic electronics. J. Mater. Chem. C 2016, 4, 3915–3933. https://doi.org/10.1039/C5TC04390E.
- Nakanishi, T. (Ed.) Functional Organic Liquids; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. i–xii.
- Ghosh, A.; Nakanishi, T. Frontiers of solvent-free functional molecular liquids. Chem. Commum. 2017, 53, 10344–10357. https://doi.org/10.1039/C7CC05883G.
- Tateyama, A.; Nakanishi, T. Responsive molecular liquid materials. Responsive Mater. 2023, 1, e20230001. https://doi.org/10.1002/rpm.20230001.
- Lu, F.; Nakanishi, T. Solvent-Free Luminous Molecular Liquids. Adv. Opt. Mater. 2019, 7, 1900176. https://doi.org/10.1002/adom.201900176.
- Wakchaure, V.C.; Channareddy, G.; Babu, S.S. Solvent-Free Organic Liquids: An Efficient Fluid Matrix for Unexplored Functional Hybrid Materials. Acc. Chem. Res. 2024, 57, 670–684. https://doi.org/10.1021/acs.accounts.3c00670.
- Tateyama, A.; Nagura, K.; Yamanaka, M.; Nakanishi, T. Alkyl–π Functional Molecular Gels: Control of Elastic Modulus and Improvement of Electret Performance. Angew. Chem. Int. Ed. 2024, 63, e202402874. https://doi.org/10.1002/anie.202402874.
- Omura, T.; Morisako, S.; Isoda, K. Amino acid-appended pyromellitic diimide liquid materials, their photoluminescence, and thermal response turning photoluminescence off. Chem. Commum. 2024, 60, 9352–9355. https://doi.org/10.1039/D4CC02229G.
- Ogoshi, T.; Azuma, S.; Wada, K.; Tamura, Y.; Kato, K.; Ohtani, S.; Kakuta, T.; Yamagishi, T.-A. Exciplex Formation by Complexation of an Electron-Accepting Guest in an Electron-Donating Pillar [5] arene Host Liquid. J. Am. Chem. Soc. 2024, 146, 9828–9835. https://doi.org/10.1021/jacs.3c14582.
- Xu, Z.; Wang, Z.; Yao, W.; Gao, Y.; Li, Y.; Shi, H.; Huang, W.; An, Z. Supercooled Liquids with Dynamic Room Temperature Phosphorescence Using Terminal Hydroxyl Engineering. Angew. Chem. Int. Ed. 2023, 62, e202301564. https://doi.org/10.1002/anie.202301564.
- Tanabe, Y.; Tsutsui, H.; Matsuda, S.; Shikita, S.; Yasuda, T.; Isoda, K. Pyromellitic-Diimide-Based Liquid Material Forming an Exciplex with Naphthalene. ChemPhotoChem 2023, 7, e202200287. https://doi.org/10.1002/cptc.202200287.
- Sato, Y.; Mutoh, Y.; Morishita, S.; Tsurumachi, N.; Isoda, K. Stimulus-Responsive Supercooled π-Conjugated Liquid and Its Application in Rewritable Media. J. Phys. Chem. Lett. 2021, 12, 3014–3018. https://doi.org/10.1021/acs.jpclett.1c00247.
- Ikenaga, A.; Akiyama, Y.; Ishiyama, T.; Gon, M.; Tanaka, K.; Chujo, Y.; Isoda, K. Stimuli-Responsive Self-Assembly of π-Conjugated Liquids Triggers Circularly Polarized Luminescence. ACS Appl. Mater. Interfaces 2021, 13, 47127–47133. https://doi.org/10.1021/acsami.1c13119.
- Isoda, K.; Matsubara, M.; Ikenaga, A.; Akiyama, Y.; Mutoh, Y. Reversibly/irreversibly stimuli-responsive inks based on N-heteroacene liquids. J. Mater. Chem. C 2019, 7, 14075–14079. https://doi.org/10.1039/C9TC05195C.
- Isoda, K.; Ishiyama, T.; Mutoh, Y.; Matsukuma, D. Stimuli-Responsive Room-Temperature N-Heteroacene Liquid: In Situ Observation of the Self-Assembling Process and Its Multiple Properties. ACS Appl. Mater. Interfaces 2019, 11, 12053–12062. https://doi.org/10.1021/acsami.8b21695.
- Giri, N.; Del Pópolo, M.G.; Melaugh, G.; Greenaway, R.L.; Rätzke, K.; Koschine, T.; Pison, L.; Gomes, M.F.C.; Cooper, A.I.; James, S.L. Liquids with permanent porosity. Nature 2015, 527, 216–220. https://doi.org/10.1038/nature16072.
- Chung, K.; Kwon, M.S.; Leung, B.M.; Wong-Foy, A.G.; Kim, M.S.; Kim, J.; Takayama, S.; Gierschner, J.; Matzger, A.J.; Kim, J. Shear-Triggered Crystallization and Light Emission of a Thermally Stable Organic Supercooled Liquid. ACS Cent. Sci. 2015, 1, 94–102. https://doi.org/10.1021/acscentsci.5b00091.
- Clarke, C.J.; Tu, W.-C.; Levers, O.; Bröhl, A.; Hallett, J.P. Green and Sustainable Solvents in Chemical Processes. Chem. Rev. 2018, 118, 747–800. https://doi.org/10.1021/acs.chemrev.7b00571.
- Horváth, I.T. Introduction: Sustainable Chemistry. Chem. Rev. 2018, 118, 369–371. https://doi.org/10.1021/acs.chemrev.7b00721.
- Ghosh, A.; Yoshida, M.; Suemori, K.; Isago, H.; Kobayashi, N.; Mizutani, Y.; Kurashige, Y.; Kawamura, I.; Nirei, M.; Yamamuro, O.; et al. Soft chromophore featured liquid porphyrins and their utilization toward liquid electret applications. Nat. Commun. 2019, 10, 4210. https://doi.org/10.1038/s41467-019-12249-8.
- Goudappagouda; Manthanath, A.; Wakchaure, V.C.; Ranjeesh, K.C.; Das, T.; Vanka, K.; Nakanishi, T.; Babu, S.S. Paintable Room-Temperature Phosphorescent Liquid Formulations of Alkylated Bromonaphthalimide. Angew. Chem. Int. Ed. 2019, 58, 2284–2288. https://doi.org/10.1002/anie.201811834.
- Santhosh Babu, S.; Aimi, J.; Ozawa, H.; Shirahata, N.; Saeki, A.; Seki, S.; Ajayaghosh, A.; Möhwald, H.; Nakanishi, T. Solvent-Free Luminescent Organic Liquids. Angew. Chem. Int. Ed. 2012, 51, 3391–3395. https://doi.org/10.1002/anie.201108853.
- Babu, S.S.; Hollamby, M.J.; Aimi, J.; Ozawa, H.; Saeki, A.; Seki, S.; Kobayashi, K.; Hagiwara, K.; Yoshizawa, M.; Möhwald, H.; et al. Nonvolatile liquid anthracenes for facile full-colour luminescence tuning at single blue-light excitation. Nat. Commun. 2013, 4, 1969. https://doi.org/10.1038/ncomms2969.
- Wakchaure, V.C.; Pillai, L.V.; Goudappagouda; Ranjeesh, K.C.; Chakrabarty, S.; Ravindranathan, S.; Rajamohanan, P.R.; Babu, S.S. Charge transfer liquid: A stable donor–acceptor interaction in the solvent-free liquid state. Chem. Commum. 2019, 55, 9371–9374. https://doi.org/10.1039/C9CC03671G.
- Iguchi, H.; Furutani, H.; Kimizuka, N. Ionic Charge-Transfer Liquid Crystals Formed by Alternating Supramolecular Copolymerization of Liquid π-Donors and TCNQ. Front. Chem. 2021, 9, 657246. https://doi.org/10.3389/fchem.2021.657246.
- Al Kobaisi, M.; Bhosale, S.V.; Latham, K.; Raynor, A.M.; Bhosale, S.V. Functional Naphthalene Diimides: Synthesis, Properties, and Applications. Chem. Rev. 2016, 116, 11685–11796. https://doi.org/10.1021/acs.chemrev.6b00160.
- Bhosale, S.V.; Jani, C.H.; Langford, S.J. Chemistry of naphthalene diimides. Chem. Soc. Rev. 2008, 37, 331–342. https://doi.org/10.1039/B615857A.
- Langford, S.J.; Latter, M.J.; Woodward, C.P. Progress in Charge Transfer Systems Utilizing Porphyrin Donors and Simple Aromatic Diimide Acceptor Units. Photochem. Photobiol. 2006, 82, 1530–1540. https://doi.org/10.1111/j.1751-1097.2006.tb09808.x.
- Wang, Y.; Wu, H.; Stoddart, J.F. Molecular Triangles: A New Class of Macrocycles. Acc. Chem. Res. 2021, 54, 2027–2039. https://doi.org/10.1021/acs.accounts.1c00108.
- Hartmann, D.; Penty, S.E.; Zwijnenburg, M.A.; Pal, R.; Barendt, T.A. A Bis-Perylene Diimide Macrocycle Chiroptical Switch. Angew. Chem. Int. Ed. 2025, 64, e202501122. https://doi.org/10.1002/anie.202501122.
- Tominaga, M.; Kawahata, M.; Itoh, T.; Yamaguchi, K. Spherical Aggregates and Crystal Structure of Naphthalenediimide-Based Macrocycle and Complexation with Perylene. Cryst. Growth Des. 2018, 18, 37–41. https://doi.org/10.1021/acs.cgd.7b01361.
- Beldjoudi, Y.; Narayanan, A.; Roy, I.; Pearson, T.J.; Cetin, M.M.; Nguyen, M.T.; Krzyaniak, M.D.; Alsubaie, F.M.; Wasielewski, M.R.; Stupp, S.I.; et al. Supramolecular Tessellations by a Rigid Naphthalene Diimide Triangle. J. Am. Chem. Soc. 2019, 141, 17783–17795. https://doi.org/10.1021/jacs.9b08758.
- Ling, Q.-H.; Zhu, J.-L.; Qin, Y.; Xu, L. Naphthalene diimide- and perylene diimide-based supramolecular cages. Mater. Chem. Front. 2020, 4, 3176–3189. https://doi.org/10.1039/D0QM00540A.
- Jhulki, S.; Feriante, C.H.; Mysyk, R.; Evans, A.M.; Magasinski, A.; Raman, A.S.; Turcheniuk, K.; Barlow, S.; Dichtel, W.R.; Yushin, G.; et al. A Naphthalene Diimide Covalent Organic Framework: Comparison of Cathode Performance in Lithium-Ion Batteries with Amorphous Cross-linked and Linear Analogues, and Its Use in Aqueous Lithium-Ion Batteries. ACS Appl. Energy Mater. 2021, 4, 350–356. https://doi.org/10.1021/acsaem.0c02281.
- van der Jagt, R.; Vasileiadis, A.; Veldhuizen, H.; Shao, P.; Feng, X.; Ganapathy, S.; Habisreutinger, N.C.; van der Veen, M.A.; Wang, C.; Wagemaker, M.; et al. Synthesis and Structure–Property Relationships of Polyimide Covalent Organic Frameworks for Carbon Dioxide Capture and (Aqueous) Sodium-Ion Batteries. Chem. Mater. 2021, 33, 818–833. https://doi.org/10.1021/acs.chemmater.0c03218.
- Wang, L.-L.; Ni, X.-Q.; Han, Y.-J.; Zhang, J.; Luo, H.-B.; Qiao, Q.; Wu, Y.-P.; Ren, X.-M. Acidified naphthalene diimide covalent organic frameworks with superior proton conduction for solid-state proton batteries. J. Mater. Chem. C 2025, 13, 4398–4404. https://doi.org/10.1039/D4TC04322G.
- Huang, Z.; Zhang, Y.; Zhao, S.; Xu, Y.; Qi, X.; Zhang, L.; Zhao, Y. Two-dimensional covalent organic frameworks with spatial-distribution defined D-A structures for efficient near-infrared photothermal conversion. Microporous Mesoporous Mater. 2022, 343, 112191. https://doi.org/10.1016/j.micromeso.2022.112191.
- Katz, H.E.; Johnson, J.; Lovinger, A.J.; Li, W. Naphthalenetetracarboxylic Diimide-Based n-Channel Transistor Semiconductors: Structural Variation and Thiol-Enhanced Gold Contacts. J. Am. Chem. Soc. 2000, 122, 7787–7792. https://doi.org/10.1021/ja000870g.
- Katz, H.E.; Lovinger, A.J.; Johnson, J.; Kloc, C.; Siegrist, T.; Li, W.; Lin, Y.Y.; Dodabalapur, A. A soluble and air-stable organic semiconductor with high electron mobility. Nature 2000, 404, 478–481. https://doi.org/10.1038/35006603.
- He, T.; Stolte, M.; Burschka, C.; Hansen, N.H.; Musiol, T.; Kälblein, D.; Pflaum, J.; Tao, X.; Brill, J.; Würthner, F. Single-crystal field-effect transistors of new Cl2-NDI polymorph processed by sublimation in air. Nat. Commun. 2015, 6, 5954. https://doi.org/10.1038/ncomms6954.
- Kao, C.-C.; Lin, P.; Shen, Y.-Y.; Yan, J.-Y.; Ho, J.-C.; Lee, C.-C.; Chan, L.-H. Solid-state structure of the naphthalene-based n-type semiconductor, and performance improved with Mo-based source/drain electrodes. Synth. Met. 2008, 158, 299–305. https://doi.org/10.1016/j.synthmet.2008.01.019.
- Basak, S.; Nandi, N.; Paul, S.; Banerjee, A. Luminescent Naphthalene Diimide-Based Peptide in Aqueous Medium and in Solid State: Rewritable Fluorescent Color Code. ACS Omega 2018, 3, 2174–2182. https://doi.org/10.1021/acsomega.7b01813.
- Sakai, N.; Mareda, J.; Vauthey, E.; Matile, S. Core-substituted naphthalenediimides. Chem. Commum. 2010, 46, 4225–4237. https://doi.org/10.1039/C0CC00078G.
- Yuan, W.Z.; Lu, P.; Chen, S.; Lam, J.W.Y.; Wang, Z.; Liu, Y.; Kwok, H.S.; Ma, Y.; Tang, B.Z. Changing the Behavior of Chromophores from Aggregation-Caused Quenching to Aggregation-Induced Emission: Development of Highly Efficient Light Emitters in the Solid State. Adv. Mater. 2010, 22, 2159–2163. https://doi.org/10.1002/adma.200904056.
- Pervin, R.; Manian, A.; Chen, Z.; Christofferson, A.J.; Owyong, T.C.; Bradley, S.J.; White, J.M.; Ghiggino, K.P.; Russo, S.P.; Wong, W.W.H. Medium effects on the fluorescence of Imide-substituted naphthalene diimides. J. Photochem. Photobiol. A 2023, 436, 114364. https://doi.org/10.1016/j.jphotochem.2022.114364.
- Hong, Y.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission. Chem. Soc. Rev. 2011, 40, 5361–5388. https://doi.org/10.1039/C1CS15113D.
- Molla, M.R.; Ghosh, S. Aqueous self-assembly of chromophore-conjugated amphiphiles. Phys. Chem. Chem. Phys. 2014, 16, 26672–26683. https://doi.org/10.1039/C4CP03791J.
- Lasitha, P.; Prasad, E. Orange red emitting naphthalene diimide derivative containing dendritic wedges: Aggregation induced emission (AIE) and detection of picric acid (PA). RSC Adv. 2015, 5, 41420–41427. https://doi.org/10.1039/C5RA04857E.
- Mollick, S.; Mukherjee, S.; Kim, D.; Qiao, Z.; Desai, A.V.; Saha, R.; More, Y.D.; Jiang, J.; Lah, M.S.; Ghosh, S.K. Hydrophobic Shielding of Outer Surface: Enhancing the Chemical Stability of Metal–Organic Polyhedra. Angew. Chem. Int. Ed. 2019, 58, 1041–1045. https://doi.org/10.1002/anie.201811037.
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; et al. Gaussian 16, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2019.
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. https://doi.org/10.1002/jcc.21759.
- Bauernschmitt, R.; Ahlrichs, R. Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem. Phys. Lett. 1996, 256, 454–464. https://doi.org/10.1016/0009-2614(96)00440-X.
- Casida, M.E.; Jamorski, C.; Casida, K.C.; Salahub, D.R. Molecular excitation energies to high-lying bound states from time-dependent density-functional response theory: Characterization and correction of the time-dependent local density approximation ionization threshold. J. Chem. Phys. 1998, 108, 4439–4449. https://doi.org/10.1063/1.475855.
- Yanai, T.; Tew, D.P.; Handy, N.C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. https://doi.org/10.1016/j.cplett.2004.06.011.
- Chirico, R.D.; Knipmeyer, S.E.; Nguyen, A.; Steele, W.V. The thermodynamic properties to the temperature 700 K of naphthalene and of 2,7-dimethylnaphthalene. J. Chem. Thermodyn. 1993, 25, 1461–1494. https://doi.org/10.1006/jcht.1993.1148.
- Okamoto, N.; Oguni, M. Discovery of crystal nucleation proceeding much below the glass transition temperature in a supercooled liquid. Solid State Commun. 1996, 99, 53–56. https://doi.org/10.1016/0038-1098(96)00139-1.
- Dai, J.; McKee, M.L.; Samokhvalov, A. Adsorption of naphthalene and indole on F300 MOF in liquid phase by the complementary spectroscopic, kinetic and DFT studies. J. Porous Mater. 2014, 21, 709–727. https://doi.org/10.1007/s10934-014-9818-3.
- Berlman, I.B.; Weinreb, A. On the fluorescence spectrum and decay time of naphthalene. Mol. Phys. 1962, 5, 313–319. https://doi.org/10.1080/00268976200100351.





